Top Industry Players in the Bone Void Fillers Market
Nearly 10 million individuals above the age of 50 suffer from hip, spine, or wrist fractures at some point in life. An additional 33.6 million individuals develop low bone mass and are at risk of osteoporosis. The growing number of complications related to bone-associated disorders such as fractures, traumas, bone infections, and bone tumors has led to the increasing demand for bone void fillers. The bone void fillers have become an indispensable tool for surgeons to manage and treat bone disorders. With the growing prevalence of bone-related diseases and accidents, many leading companies are introducing innovative solutions to provide patients with high-quality bone void fillers to achieve the best outcomes.
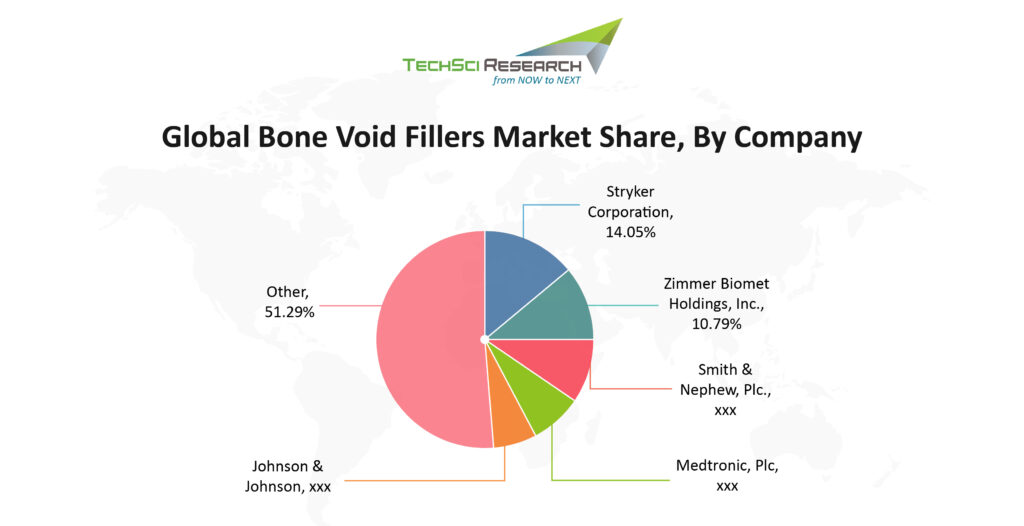
Here are the top ten companies with the majority of market share in the global bone void filler market.
Stryker Corporation
Headquarters: United States
Establishment: 1941
Bone Void Filler Product Portfolio: Vikos void filler, BIO chips, Demineralized Bone Matrix (DBM), Viable Bone Matrix (VBM), Vitoss Bone Matrix, BIO DBM, and many more.
One of the leading companies in medical technology, Stryker Corporation deals in innovative products and services related to orthopedics, medical, surgical, and neurotechnology. The company has been expanding its range of orthopedic products for knees, trauma, extremities, and hips to address various requirements of patients. The bone void fillers are available in different sizes and forms, such as gel, putty, Putty combined with cancellous chips. Stryker Corp. provides its products in 36 countries, including developed and developing countries. Strong distribution networks and new product launches are some of the strategies adopted by Stryker to become a market leader in the global bone void filler market.
Zimmer Biomet Holdings
Headquarters: United StatesEstablishment: 1927
Bone Void Filler Product Portfolio: Calcigen S, CopiOs, γGamma-bsm Moldable PuttyBone Substitute Material, EquivaBone, Stagraft DBM, GPS III, Plasmax, BIOCUE, Bonus Traid allograft, and others.
Zimmer Biomet Holdings is a leader in musculoskeletal healthcare, providing a range of orthopedic reconstructive products, including biologics, extremities and trauma products, spine, dental implants, etc. The company has a distribution network across 40 countries, covering major geographics in the United States, Europe, the Middle East, Asia-Pacific, and Africa. Continuous innovation and new product launches have supported the company’s growth over the years. Besides, Zimmer Biomet Holdings has continuously expanded its distribution network globally with mergers and acquisitions. Till 2020, Zimmer had acquired 19 companies to strengthen its position in the global market further.
Smith & Nephew Plc
Headquarters: United Kingdom
Establishment: 1856
Bone Void Filler Product Portfolio: Graftys HBS, Graftys Quick Set, and Strucsure CP
Leading medical equipment manufacturing firm, Smith & Nephew operates majorly in three healthcare segments including Orthopedics, sports medicine & ENT and advance wound management. The orthopedic segment is further divided into knee implants, hip implants, trauma, and other reconstruction. Smith & Nephew has signed a deal with Graftys, which gives them distribution rights to sell a unique bone graft technology that offers surgeons fast-setting, macroporous calcium phosphate bone void filler to provide support for bone growth. The company operates in 100 different countries, including major geographics like United States, Europe, Middle East & Africa, and Asia Pacific.
Medtronic
Headquarters: Ireland
Establishment: 1949
Bone Void Filler Product Portfolio: Grafton crunch DBM, Corex, Grafton putty, Grafton Orthoblend, Grafton gel, and Progenix
A Fortune 500 company with incredible portfolio of integrated medical solutions, Medtronic has its products segmented across four business units, such as cardiac and vascular group, restorative therapies, Diabetes treatments, and minimally invasive therapies. The healthcare giant is the only company to combine robotics, biologics, navigation, and spinal implants. In 2019, Medtronic introduced Grafton demineralized bone matrix (DBM) bone graft product, a first-of-its-kind product in the Japanese market, which revolutionized the bone void filler market. Medtronics is currently operating more than 150 different countries, including the United States, emerging market, and non-US developed markets.
Johnson & Johnson
Headquarters: United States
Establishment: 1886
Bone Void Filler Product Portfolio: DBX Demineralized Bone Matrix (CMF), Norian drillable (Biocompatible bone void filler), Vertecem II bone cement, ChronOS bone graft substitute, Vivigen cellular allograft
Johnson & Johnson is a well-established player in the orthopedics segment and offers a broad range of products for hips, knees, trauma, spine, and others. DePuy Synthes, a subsidiary of Johnson & Johnson, manufactures, markets, and distributes orthopedic products designed for damaged joints, bone repairing, and reconstruction of traumatic skeletal injuries in different sizes and forms like paste, mix, injectable, and putty. J&J’s strong presence in more than 60 countries and diverse product portfolio of synthetic and biological bone void fillers contributes to the company’s greater share in the global bone void filler market.
BoneSupport AB
Headquarters: Sweden
Establishment: 2000
Bone Void Filler Product Portfolio: Cerament Bone Void Filler, Cerament G, Cerament V
BoneSupport AB is one of the fastest-growing companies in the orthobiologics segment that provides a wide range of new and advanced products to treat bone disorders, revision arthroplasty, chronic osteomyelitis, and different ankle surgeries. The company started operating in the bone void fillers segment in 2009 with CE-Mark for Cerament bone void fillers. In 2012, BoneSupport AB entered the US market and now operates in the United Kingdom, Germany, Sweden, Denmark, Switzerland, Spain, Netherlands, and France. In March 2020, the US-FDA recognized Cerament G as a breakthrough medical device for the indication of osteomyelitis and aimed to reach a potential approval for bone infection in 2022.
NuVasive, Inc.
Headquarters: United States
Establishment: 1997
Bone Void Filler Product Portfolio: Attrax bone void filler, Osteocel Plus bone graft, Osteocel Pro bone graft, Propel DBM Putty and gel
NuVasive is one of the fastest-growing companies in terms of spine surgery and integrated solutions. The company mainly operates in two business segments: spinal hardware and surgical support (intraoperative neuromonitoring services, disposable, and biologics). The company offers Demineralized Bone Matrix Putty and Gel, widely used in extremities, spine, and pelvis. NuVasive’s bone void fillers have demonstrated improved performance, such as increased bone formation and mechanically stronger fusions. In Jan 2022, FDA cleared NuVasive’s bone void fillers for the expanded usage of Attrax Putty in interbody fusions of the thoracolumbar spine. The company operates in 50 countries, but most of its revenue comes from the United States.
Integra LifeSciences Corporation
Headquarters: United States
Establishment: 1989
Bone Void Filler Product Portfolio: Integra OS, Trel-X, Trel-XC, Accell Evo 3, Orthoblast, Mozaik, Dyna Graft, and many others.
Integra LifeSciences is a global leader in regenerative and neurosurgical technologies and extremity orthopedic solutions dedicated to providing the best patient care. Integra LifeSciences offers a comprehensive portfolio of high-quality products and services, mainly in two business segments such as Codman specialty surgical and orthopedics & tissue technologies. The Mozaik Moldable Morsels bone void fillers are available in different sizes to fill the gaps in voids in the spine, pelvis, or skeletal system. Besides, the company has extended its product portfolio by launching new and small size bone void fillers, which can be used in smaller amounts.
Arthrex Inc.
Headquarters: United States
Establishment: 1981
Bone Void Filler Product Portfolio: Quickset Calcium Phosphate Cement, INNOTERE Paste-CPC and INNOTERE 3D Scaffolds
Pioneer in the field of arthroplasty and orthobiologics, Arthrex develops more than 1000 innovative products and procedures every year. The company offers a range of orthobiologics products such as bone, cartilage, and cellular products, soft tissue and viscosupplementation, bone repair kits, and bone void fillers. The bone void fillers provided by Arthrex are synthetic and bioabsorbable, which are produced through the 3D printing process. Additionally, the company offers a Global Enterprise Labeling System (GELS) to provide a streamlined approach for managing products and product-related data. The major geographic areas covered by Arthrex are the United States, Europe, Middle East, Asia-Pacific, Latin America, and Western Hemisphere.
Collagen Matrix Inc.
Headquarters: United States
Establishment: 1997
Bone Void Filler Product Portfolio: Ossimend Strips, Ossimend Block, OssiMend Putty, Tenomend, Guidor, SynOss Plug, SynOss Putty, and many more.
Collagen Matrix is collagen and mineral based medical device manufacturer operating in five business segments such as dental, spine, orthopedic, dural repair, and nerve repair. The bone void fillers provided by the company are highly purified, biocompatible matrix, and is osteoconductive. Ossimend blocks are good to handle while they are wet, OssiMend Putty supports body’s natural ability to regenerate bones, and Guidor fills defects of various sizes when applied directly to the targeted area. In 2019, Collagen Matrix was occupied by the Chicago-based healthcare private firm, Linden Capital Partners. Strategic collaborations with companies across the globe for strengthening distribution channels, contract product development, and manufacturing have contributed to a significant share of the company in the global bone void filler market.
Many new healthcare companies are also venturing into the orthopedic market, which could lead to the expansion of more quality products, which could help in further improving patient outcomes.
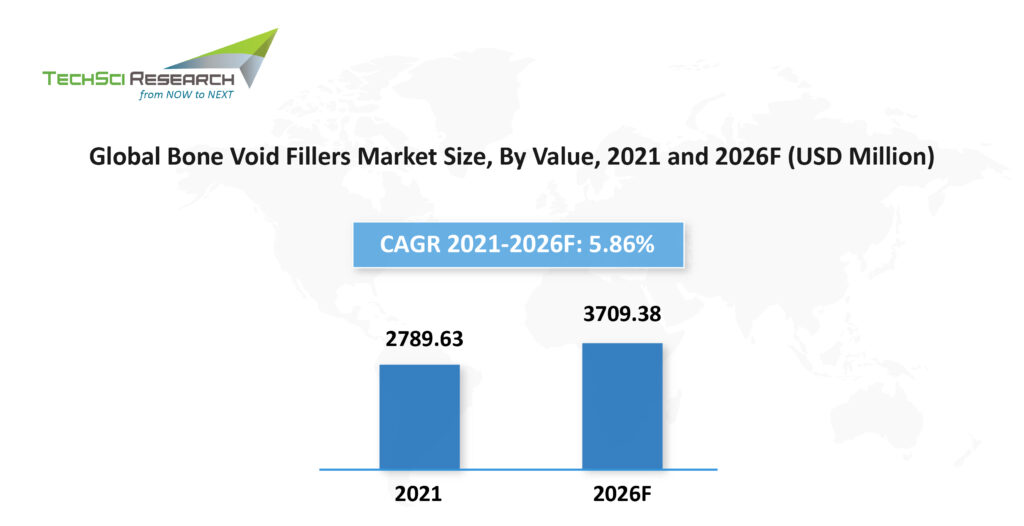
According to TechSci Research report on “Bone Void Fillers Market – Global Industry Size, Share, Trends, Competition, Opportunity, and Forecast, 2016-2026 Segmented By Material (Calcium Sulphate, Demineralized Bone Matrix, Tri-Calcium Phosphate, Others), By Form (Putty, Paste, Granules, Gel, Others), By Procedure (Spine Fusion & Interbody Fusion, Oral Surgeries, Others), By End User (Hospitals, Specialty Clinics, Others), By Region”, the global bone void fillers market is expected to register a steady CAGR of 5.86% during the forecast period. The factors attributing to the growth are increasing incidences of osteoporosis, spinal cord injuries, and accidents, etc. Besides, the growing prevalence of diabetes that leads to osteomyelitis is positively impacting the growth of global bone void fillers market.